The Mole 1 dozen = 1 century= 1 pair = 1 mole = x There are exactly 12 grams of carbon-12 in one mole of carbon ppt download

The Mole Standards 1 dozen = 1 gross = 1 ream = 1 mole = x There are exactly 12 grams of carbon-12 in one mole of carbon ppt download

The Mole Standards 1 dozen = 1 gross = 1 ream = 1 mole = x There are exactly 12 grams of carbon-12 in one mole of carbon ppt download

Chemistry Warm Up: Mole / Mass / Particles 1.What is the mass of one mole of water? 2.If one milliliter of water has a mass of 1.00grams, how many moles. - ppt download

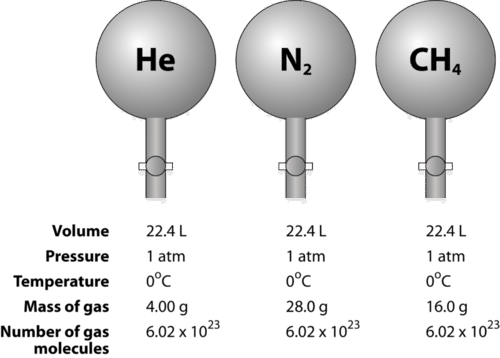
The Mole What's a mole? In chemistry, a mole is a counting unit. What does that mean? –1–1 dozen = 12 units(ie eggs,donuts) –1–1 mole = 6.02x10 23 particles. - ppt download